Types of Chemical Reactions
There are many types of chemical reactions. Five of the most common are:
synthesis: two or more reactants combine to form a single product.
A + B à C
decomposition: one reactant disintegrates (decomposes) to form two or more products:
A à B + C
single replacement (sometimes called single displacement): atoms of one element replace atoms of another in a compound:
A + BC à AC + B
Most often, AC and BC are ionic compounds, which means A and B are metals, and C is a non-metal or negative polyatomic ion.
As an analogy, imagine that BC are a couple, and C breaks up with B to go out with A.
double replacement (sometimes called double displacement): when two positive ions (or two negative ions) switch with each other to form two new compounds:
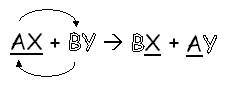
As an analogy, imagine that AX and CY are two couples. A and B switch boyfriends, so B is now going out with X and A is now going out with Y.
combustion: a special kind of reaction in which a hydrocarbon (a compound containing carbon and hydrogen) reacts with O2 (burns, or “combusts”) to form CO2 and H2O. For example:
C3H8 (ℓ) + 5 O2 (g) à 3 CO2 (g) + 4 H2O (g) + heat
The internal combustion engine in your car is a special reactor in which octane (C8H18) combusts in a cylinder, producing heat. The heat makes the gases inside the cylinder expand. (Remember the gas laws!) This pushes the piston down, which makes the car go.